AbstractMalaria is by far the global health problem with more than 225 million infected cases and 781 thousand deaths in 108 countries (WHO malaria report, 2010).The causative agents in humans are four recognized species of malaria parasites, including P.falciparum, P.vivax, P.malaria and P.ovale. P. falciparum is prevalent in tropical Africa, Asia and Latin America. The distribution of P. vivax is worldwide, mostly in temperate regions.P. ovale infections are seldom seen in all geographical areas of the malaria-endemic world, mainly in tropical west Africa and on some islands of the western Pacific. Of the four human Plasmodia species reported in Vietnam, P.falciparum is the most prevalent, accounting for nearly 80%, P.vivax contributing around 15-20% and P.malariae uncommon with only less than 30 cases recorded from Central areas and West Highlands in 2010. To P.ovale infection, there were 4 documented cases in US residents returning from Vietnam in 1969 (by microscope and IFA) and 3 cases recorded in 1995 (by AO and PCR). However, up to now, P.ovale infections cases that were detected under microscopy are especially rare in Vietnam.
This is one P.ovale infection case which was detected by Giemsa microscopy, i.e. staining blood film with Giemsa and morphologically diagnosing under an optical microscope.
1. Background:
Plasmodium ovale has been reportedly found in some areas of the world; however, according to Lysenko and Bejaev, the natural distribution of P.ovale is mainly in sub-Saharan Africa and some islands of the Western Pacific. According to Bruce Chwatt (1963), Gill and Warell (3rd Essential Malariology, 1963) malaria mosquitoes transmit P.ovale mostly in West Africa. Medical literature show that P.ovale is found sporadically in Southeast Asia countries. In Vietnam, P.falciparum is the most popular parasite, next is P.vivax; P. ovale contributes to only a small number of malaria infections, scatteredly distributing in some provinces of Central Vietnam. In the coastal and highland provinces of Central Vietnam, according to the report system from 1976 - 2010, only one case of P.ovale infection was documented in Dak Lak province (1995).
The studies conducted in Vietnam have confirmed 4 cases of infection with P.ovale malaria among American soliders who were garrisoned in the South of Vietnam from January 1966 to March 1969 (Gleason N.N). Recently, Kawamoto. F carried out his researches in 4 provinces (Song Be, Lam Dong, Dak Lak and Khanh Hoa) from 1994 - 1995 by Acridine Orange (AO) Stainingfor rapid diagnosis and hybrid technique based on DNA amplification assay, and the results showed 3 cases infected with P.ovale. In 1996, Le Duc Dao reported the presence of P.ovale (by PCR technique) in Khanh Vinh district, Khanh Hoa province. The objective of this report is to describe one case of P.ovale infection detected with Giemsa staining and examined under light microscopy.
2. Object and method of study:
2.1. Object of study: Malaria parasites in thick and thin blood smears
2.2. Method of study: Describe the morphology of malaria parasites and take their digital photographs through a light microscope
Giemsa-stained thin and thick blood films |
3. Results of the description of one P.ovale infection case in Binh Thuan province
The patient was male, 27-year-old, frequently worked in the forest, went to the communal health station of Phan Son, Bac Binh district, Binh Thuan province (2008) with the symptoms of fever and chills. Before that, this patient had had fever every other day, chills, right lower quadrant pain, myalgia, vomitting. He was treated with chloroquin for 3 days (4,2,2) and primaquine with 4 tablets per day x 10 days. After treatment, the patient was free from fever; yet medical staff couldn't monitor him then.
The figure of P.ovale in thin blood smear:
The figure 1: Mature Trophozoite:
The nuclei were purple, firm and round in shape. The blue protoplasm developed around the nuclei. A varying amount of pigment, dark brown in colour, and disposed in scattered grains on the protoplasm. The pink coarse Shuffner's dots were scattered through red cell. The parasitized red cells were not enlarged, oval-shaped and pointed at one end.
Figure 2: Mature trophozoite:
The nuclei were firm, purple, three-leafed- shaped. The protoplasm was delicate, blue and extended from both sides of the nucleus. Few dark brown pigments were scattered on the protoplasm. The pink coarse Shuffner's dots were scattered through the erythrocyte. The parasitized erythrocytes were stretched into oval shape with fimbriation at two stretched sides.
Figure 3: Gametocyte
Thenucleus purple, extendedandlocated at the margin of the protoplasm. The protoplasm was thick, slightly blue, filling most of the erythrocyte. The golden brown pigment-granules disposed scatteredly through the protoplasm. The rough pink Schuffner's dots were scattered through the erythrocyte. The parasitized red blood cells was pyriform.
Figure 4 (P101012, Schizonte:
There were 6 reddish nucleic whose edges and sizes are irregular. The protoplasm was bluish, and has not been divided, filled about two third of the erythrocyte. The golden brown pigment-granules gathered at the edge of the protoplasm. The Shüffner's dots were coarse, visible and pink, scattered throughout the erythrocyte. The parasitized erythrocytes were oval in shape and fimbriated atthe top edge.
Figure 5 (P1010019): two parasites on a microscopic scale
The left parasite, Schizonte:
There were 8 pink nucleic whose sizes are irregular. The protoplasm was bluish, and not divided,filled about two third of the erythrocyte. The numerous golden brown pigment-granules disposed scatteredly through the protoplasm. The Shüffner's dots were coarse, scattered throughout the erythrocyte. The parasitized erythrocytes were oval-shaped.
The right parasite: Mature trophozoite:
The nucleus was round and coarse, beside the edge of the erythrocyte. The protoplasm was bluish, extended into a strip circling the nucleus. The pigment-granules were few, dark brown, clustering through the protoplasm. The Shüffner's dots were coarse, visible and pink, scattered throughout the erythrocyte. The parasitized erythrocytes are oval, and fimbriated at one end.
Most of the parasites detected on the blood smears were at the mature trophozoite, schizonte and gametocyte stages. The ring stage didnot appear on the blood smear in this case. The special characteristics were that most of the parasitized erythrocytes were oval-shaped and have fimbriation in the shape of saw-tooth or pennon. The erythrocyte was less enlarged, like in the case of P.vivax infection. The above descriptions also showed that this parasite was more compact than P.vivax; the chromosome and macronucleus occupied nearly two third of the erythrocyte. The outside edge of the parasitized erythrocyte werenot clear, the erythrocyte had a lot of magenta Shüffner's dots. The features of malaria parasite conformation described in this case is typical of P.ovale, like other authors had described (Wilcox, Jeffery, Young - 1954) and Garham (1966).
4. Discussions:
P.ovale was described by Stephen in 1922. P.ovale distributes mostly in Africa, and with a very low proportion in western Pacific countries. In Vietnam, P.ovale infections are rarely found. The development stages of the malaria parasite detected on this thin blood film is typical of P.ovale. According to the report of the health station, in a recent year, the patient often went there for medical examination and there weren't any severe symptoms that needed blood transfusion at provincial and district hospitals. But in case of blood transfusion, the symptoms also appeared after 10 days from the transfusion day. However, it could be affirmed that the patient hasn't gone abroad.
As regards the mosquito vectors of P.ovale malaria, according to William E. Collins and Geoffrey M. Jeffery, An.gambiae, An.funestus, An.atroparvus, An.albimanus, An.quadrimaculatus, A.freeborni, A. maculatus,A. subpictus; A.stephensi, A.dirus, A.farauti are likely to be infected with P.ovale and transmit it. The surveys in Phan Son commune showed that there had been the presence of An. dirus, An.minimus and An.maculatus. This is the necessary condition for the transmission of P.ovale malaria in this area. Therefore, the most convincing theory of this malaria infection case is because of the spread of malaria in the malaria-endemic area of Binh Thuan province. After certifying this case of P.ovale infection, the IMPE-QN conducted two other surveys in this area, yet no more P.ovale malaria cases were detected despite using microscopic technique and even molecular biological test. Rare are P.ovale infections that havebeen reported in Vietnam so far.
The above discussions showed that P.ovale malaria is prevalent with a very low rate in the endemic areas of Vietnam. Up to now P.ovale has been found sporadically in some provinces such as Dak Lak, Khanh Hoa, Lam Dong, Song Be.
The uncommonness of P.ovale infections makes it difficult to explain how this parasite exists in the nature. The microscopic examination of blood smear maynot sensitive enough to detect cases infected with P.ovale, whereas the infection of P.ovale often has low density and occurs intermittently. P.ovale also reduces its development as well as being hidden by other malaria parasite infections.
Furthermore, the conformation of P.ovale is probably misdiagnosed as that of P.vivax, so the microscopists may omit the detection of P.ovale conformation as examining under the optical microscopy and assume that is of P.vivax.
5. Conclusions:
The conformation of the malaria parasite on the Giemsa-stained blood film that was detected under the optical microscopy is the example of P.ovale. There has been the presence and prevalence of P.ovale in some malaria-endemic areas of South Vietnam.
6. Recommendations:
Medical technicians should prepare a thick and a thin blood film on the same slide. As examining malarial blood smears, its is neccesary to pay attention the following characteristics of P.ovale:
a) The parasitized erythrocytes are oval in shape and fimbriated at the top end; however, the oval shape and fimbriation only make up by 20-30% of infected red blood cells. The appearance of the Schuffner's dots in infected red blood cells is relevant to P.ovale infection; yet, these dots also appear in case of P.vivax infection.
b) It is difficult to differentiate the ring form of P.ovale from that of other Plasmodia species: the erythrocytes arenot enlarged and no Schüffner's dots in general.
c) Schizonte: the maximum number of merozoites are 12, fewer than P.vivax. The early stage of schizonte is similar to schizonte of P.vivax with Schuffner's dots or similar to schizonte of P.malariae without Schuffner's dots.
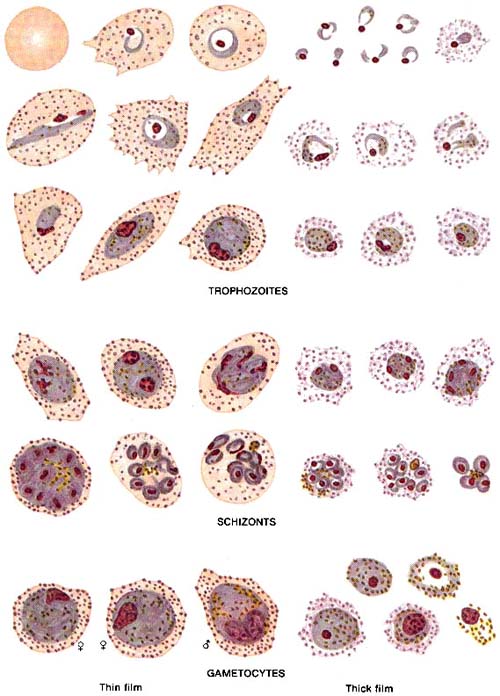 |
Appearance ofPlasmodium vivaxstages in Giemsa-stained thin and thick blood films
(The Basic Malaria Microscopy, WHO) |
References:
1. !important; Le Dinh Cong, Le Duc Dao (1996). Differential diagnosis of 4 human malaria parasites by Nested PCR technique. Information on malaria and other parasitic diseases control. The Institute of Malariology, Parasitology and Entomology Ha Noi, 1996, 04:26-33
2. !important; Alves W., Schinazi A.L., Aniceto F. 1968. P.ovaleinfections in the Philippines. Bull. W. H. O. 39:494–495.
3. !important; Baird, J. K., Purnomo, and S. Masbar. 1990. P.ovalein Indonesia. Southeast Asian J. Trop. Med. Public Health 21:541–544. (7)
4. !important; Cadigan, F. C., and R. S. Desowitz. 1969. Two cases of P.ovalefrom central Thailand. Trans. R. Soc. Trop. Med. Hyg. 63:681–682.
5. !important; Gills H.M, Warell D.A. Bruce-Chwatt’s Essential Malariology, 3rd Edition. pp.24-25.
6. !important; Gleason N.N, Fisher G.U, Blumhardt R., Roth E.A., Gaffney W.G. P.ovaleMalaria Acquired in Viet-Nam. Bull. World Health Org.1970,42, pp.399-403.
7. Hombhanje. W.F. P.ovalespecies in Papua New Guinea - lest we forget. Papua New Guinea Medical Journal Volume 41, No 3-4, Sep-Dec 1998, pp.116-118.
8. !important; Kawamoto. F, Miyake. H., Maneko. O,Kimura.M, Nguyen thi Dung, Le duc Dao. Sequence Variation in the 18S rRNA gene, a Target for PCR-Based Malaria Diagnosis, in P.ovalefrom Southern Vietnam. Journal Of Clinical Microbiology. American Society for Microbiology Sept. 1996, p. 2287–2289 Vol. 34, No. 9.
9. Lim AL.Y., Mahmud R., Chew H.C., Thiruventhiran T., Chua H.K. P.ovale infection in Malaysia: first imported case. Malaria Journal 2010, 9:272Center for Diseases Control and Prevetion. Laboratory diagnosis of malaria P.ovale.
10. !important; Lysenko.A.JA., Beljaev. A.E . 1969. An analysis of the geographical distribution of P.ovale. Bulletin. WHO. 40: pp. 383–394.